
The two children that didn’t respond to the gene therapy returned to their normal treatment regimens, and one has since received a bone marrow transplant.1Department of Biotherapy, Hôpital Necker–Enfants Malades, Assistance Publique–Hôpitaux de Paris (AP-HP), Université René Descartes, and INSERM, Centre d’Investigation Clinique intégré en Biothérapies, Groupe Hospitalier Universitaire Ouest, AP-HP, Paris, France.ĢINSERM U768, Université René Descartes, and Hôpital Necker–Enfants Malades, Paris, France.ģDepartment of Microbiology, University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania, USA.ĤInstitut Universitaire d’Hématologie (IUH), Université Denis Diderot, and Hematology Laboratory AP-HP, Hôpital St-Louis, Paris, France.ĥUnité de développement des lymphocytes, Département d’Immunologie, Institut Pasteur, and INSERM U668, Paris, France.ĦHematology Laboratory and INSERM U563, Centre de Physiopathologie de Toulouse Purpan, University Hospital Purpan, Toulouse, France.ġ0Department of Pediatric Immuno-Hematology, Hôpital Necker–Enfants Malades, AP-HP, and Université René Descartes, Paris, France.ġ1Department of Pediatric Hematology, Oncology and Clinical Immunology, Heinrich-Heine University, Düsseldorf, Germany.ġ2Department of Infectious Diseases and Immunology, University Children’s Hospital Munich, Munich, Germany.ġ3Department of Pediatrics, Louisiana State University Health Sciences Center, New Orleans, Louisiana, USA.ġ4Department of Pediatrics, Section of Immunology, University Medical Center Utrecht, Utrecht, The Netherlands.Īddress correspondence to: Salima Hacein-Bey-Abina, Hôpital Necker–Enfants Malades, 149 rue de Sèvres, F-75015 Paris, France. “All the patients are alive and well, and in more than 95% of them, the therapy appears to have corrected their underlying immune system problems,” co-lead author Donald Kohn of UCLA said in a press release. Gene therapy, using the patients own cells, avoids the need for matched donors as well as GvHD and other bone marrow transplantrelated complications. 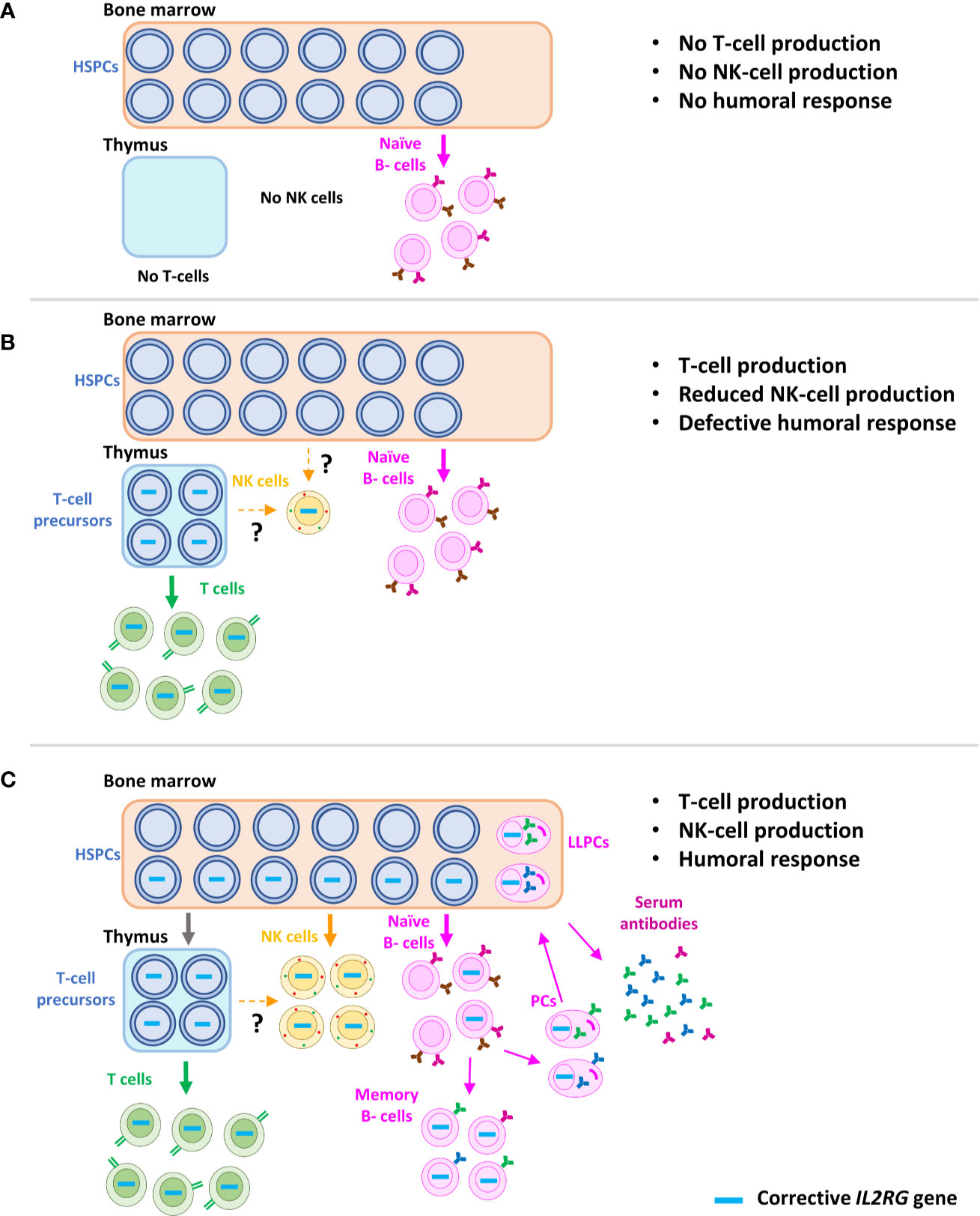
Stem cell transplants from matched, related donors can be effective, but most patients don't have such donors available to them. The researchers followed up with the children for three years, reporting the results of the trials in the New England Journal of Medicine on May 11. ADA-SCID is caused by a gene mutation that affects a critical protein called adenosine deaminase, which is needed to keep toxins in the body from destroying disease-fighting white blood cells. People ask us, is it a cure? Who knows long term, but these children are doing well. Gene Therapy Stem Cells Viral/Non-Viral Vectors Clinical-stage biopharmaceutical company Mustang Bio, has announced that the first patient in their ongoing Phase I/II clinical trial has received the ex vivo lentiviral-based gene therapy, MB-110, to treat recombinase-activating gene-1 (RAG1) severe combined immunodeficiency (RAG1-SCID). One is the direct injection of the genetic material in the blood or organ or body cavity. There are two different ways that the genetic material can be delivered. Gene therapy is the term used for any treatment where you are delivering genetic material into the body. A disabled AIDS virus was then used to inject a new copy of the ADA gene into the stem cells, which were then cycled back into the children’s bodies. Please provide a brief explanation of gene therapy for SCID. 
The gene therapy: During the new trials, blood-forming stem cells were collected from 50 children. “I just thought no one should have to suffer like this.” “I saw young bone marrow transplant patients who developed graft-versus-host disease and others who had to take all these anti-rejection medications and still had issues,” Chelsea Oakley, mother of one of the trial participants, said in a press release. Jude is driving a landmark cure for patients with X-linked severe combined immunodeficiency (SCID-X1). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
